
One millimeter of water equals 9.80665 Pascals. Inches of mercury (inHg): At 60☏, one inch of mercury equals 3376.85 Pascals.Ĭentimeter or millimeter of water (cmH 2O or mmH 2O): One centimeter of water corresponds to 98.0665 Pascals of pressure. Millimeter of mercury (mmHg): One millimeter of mercury is approximately equal to the standard atmospheric pressure divided by 760, which gives ≈ 133.322368 Pascals. We can use the depth of the liquid in the manometer as reliable pressure readings: They work on the principle of hydrostatic pressure. Units based on liquid column manometers: Liquid column manometers are common pressure measurement equipment. Standard atmospheric pressure (atm): The average atmospheric pressure at sea level is approximately equal to 101325 Pascals, which we define as the standard atmospheric pressure (atm). You can use both hPa and bar in this calculator for barometric pressure conversion. Hectopascals (hPa): Due to the advent of the SI unit system, many meteorologists use hectopascals for measuring air pressure instead of bar or millibar. In meteorology, millibars (=10 -3 bars) are common worldwide. It is slightly less than the standard atmospheric pressure at sea level (approximately 1.013 bar). One pound per square inch (1 psi) approximately equals 6895 Pascals.īar: Another common unit not part of the SI unit system, the bar is equal to 100,000 Pa (or 100 kPa). Pound per square inch (psi): Pound per square inch (or pound-force per square inch) is an avoirdupois unit of pressure caused by one pound-force acting on one square inch area. We can measure them all using Pascals, but other units are conveniently available. P = ρ 20 ∙ g ∙ h = 1.204 kg/m 3 × 9.80665 m/s 2 × 1 m = 11.There are different kinds of pressure, like fluid pressure and barometric pressure, depending on the source. Standard gravitational acceleration is g = 9.80665 m/s 2ĭensity of air at 20☌ is ρ 20 = 1.204 kg/m 3ġ m height changes the air pressure at a constant temperature of 20☌ by This law is also assumed as an air column. The hydrostatic pressure is calculated according to Blaise Pascal: 1µbar = 0.1 Pa.Ĭhange of air pressure associated with the change in height Question: How does the air pressure change if the height changes 1 meter? 1 mbar = 100 Pa = 1 hectopascal.ġ microbar is 10 −6 bar = 1 dyne/cm². Meteorologists like this.ġ millibar = 0.001 bar = 0.750 Torr. Normal air pressure = 1.01325 bar = 1013.25 mbar.ġ000 millibars are 1 bar. The bar is nearly idendic to the atmosphere unit. The pressure, which brings a collum of mercury to 1 millimeter is called 1 Torr (1 mm Hg = 1 Torr).ġ atm = 760 Torr = 14.696 psi. 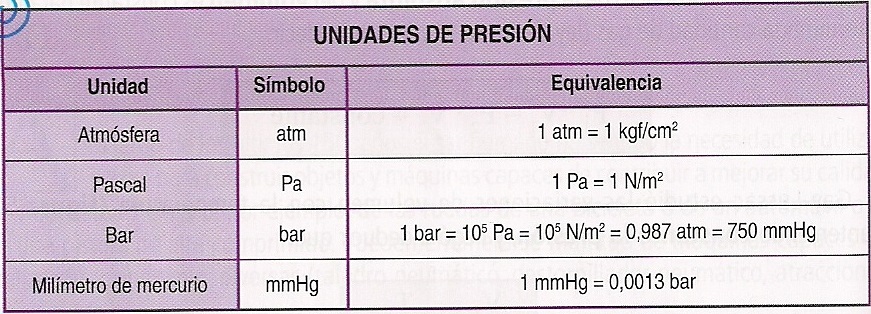
Torricelli barometer design: One atmosphere pressure brings a colums of mercury in a Hg barometer to a height of 760 millimeter. Normal atmospheric pressure is 1 atm = 14.696 psi - that means, an air colum above us of an area of 1 square inch weighs 14.696 pounds. Pressures of some gigapascal can convert graphite to diamonds or can form from hydogenium a mtallic conductor. The prefix "kilo" means "1000", therefore is 1 kilopascal = 1000 Pa and 101.325 kPa = 1 atm = 760 torr and 100 kPa = 1 bar = 750 torr. p = pressure F = force A = areaġ pascal = 10 dyne/cm 2 = 0.01 mbar. To accelerate 1 kilogram to 1 meter per second squared) 1 Pa ≡ 1 kg/(m įorce 1 N = 1 kg × m/s 2. More conversions of pressure or stress units Unitġ pascal ( p = F / A) = the force F of 1 newton per square meter area (1 newton = the force, which is used Important to know: 1 Pa = 1 N/m 2 ≡ 94 dBSPL and 1 bar = 10 5 Pa.Ĭonversion of sound units: pascals to dBSPL Standard atmospheric pressure is 101,325 pascals = 1,013.25 hPa = 101.325 kPa To the Super Calculator for pressure units You will see the program but the function will not work. 
The used browser does not support JavaScript. Right side you will get the calculated result with the adjusted unit.Ĭlick at a free space. ĭefinition: Pressure or stress = force per area, p = F / Aį = force, A = area, p = pressure in Pa = N / m 2 ≡ kg / (m Įnter on the left side the unit and the known number.Calculations and conversions of pressure units.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
